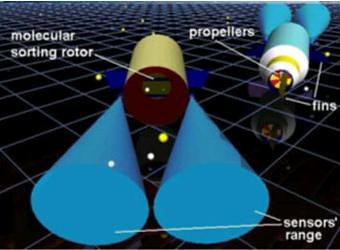
nanorobot for nanomedicine drug delivery
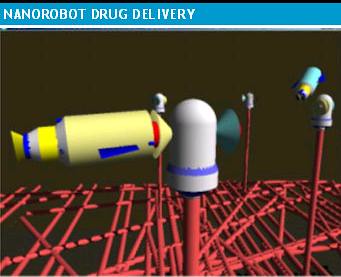
Simulated nanorobot for drug delivery
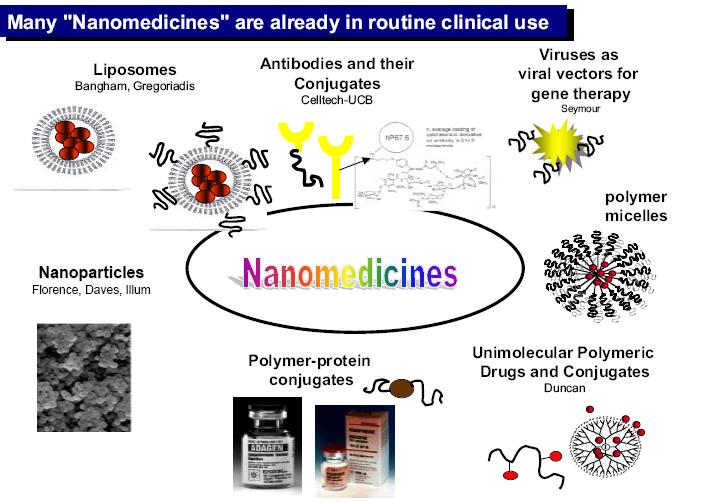
Fully operational nanorobots for biomedical instrumentation should be achieved as a result of nanobioelectronics and proteomics integration. The proposed platform should enable patient pervasive monitoring, and details are given in prognosis with nanorobots application for intracranial treatments. This integrated system also points towards precise diagnosis and smart drug delivery for cancer therapy.
The methodologies and the implemented 3D simulation described in our study served as a test bed for molecular machine prototyping. The numerical analysis and advanced simulations provided a better understanding on how nanorobots should interact inside the human body. Hence, based on such information, we have proposed the innovative hardware architecture with a nanorobot model for use in common medical applications. The nanorobot takes chemical and thermal gradient changes as interaction choices for in vivo treatments. The use of mobile phones with RF is adopted in this platform as the most effective approach for control upload, helping to interface nanorobots communication and energy supply.
The next steps in our work can be defined as follows: (a) model manufacturing with CNT-CMOS biochip integration; (b) laboratory studies for in vivo tests; and (c) commercialization. The pipeline for development in the medical sector typically requires research and efforts to get new ideas out of laboratories and into the marketplace
FURTHER READING
Nanorobot design website
They have written many papers on this work. Robert Freitas is involved in some of them.
Nanorobot architecture for medical target identification.
The nanorobot interaction with the described workspace shows how time actuation is improved based on sensor capabilities. Therefore, our work addresses the control and the architecture design for developing practical molecular machines. Advances in nanotechnology are enabling manufacturing nanosensors and actuators through nanobioelectronics and biologically inspired devices. Analysis of integrated system modeling is one important aspect for supporting nanotechnology in the fast development towards one of the most challenging new fields of science: molecular machines. The use of 3D simulation can provide interactive tools for addressing nanorobot choices on sensing, hardware architecture design, manufacturing approaches, and control methodology investigation.
Earlier work was with CMOS versions of small robots
Hardware architecture for nanorobots
Freitas' nanomedicine site
Center for Automation in Nanobiotech website